Tamsulosin Hydrochloride IP BP
Price 75000 INR/ Kilograms
MOQ : 1 Kilograms
Tamsulosin Hydrochloride IP BP Specification
- Storage
- Room Temperature
- Type
- Pharmaceutical Intermediates
- Grade
- Medicine Grade
- Physical Form
- Powder
Tamsulosin Hydrochloride IP BP Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Payment Terms
- Cash Advance (CA)
- Supply Ability
- 10 Kilograms Per Month
- Delivery Time
- 7 Days
- Main Domestic Market
- All India
About Tamsulosin Hydrochloride IP BP
Tamsulosin Hydrochloride IP BP is a kind of medication used for the treatment symptoms of an enlarged prostate gland, a condition called benign prostatic hyperplasia (BPH). It performs by relaxing the muscles in the bladder neck and prostate, allowing for easier urination. This type of medication belongs to a groups of drugs known as alpha blockers and comes in both extended release and immediate release formulations. Tamsulosin Hydrochloride IP BP is normally taken once a day, preferably at bedtime, and should be utilized with caution in patients with liver or kidney problems, low blood pressure, and those taking definite medications like nitrates.
Medicine Grade Quality Assurance
Our Tamsulosin Hydrochloride IP BP is produced to meet stringent IP (Indian Pharmacopoeia) and BP (British Pharmacopoeia) standards, ensuring exceptional purity and reliability for pharmaceutical applications. Our processes prioritize consistency and safety, making our intermediates a trusted option for manufacturing a range of medicines.
Ideal Storage and Handling
This pharmaceutical intermediate is provided in powder form, and should be stored at standard room temperature. Adherence to recommended storage conditions maintains the product's chemical integrity, which is crucial for its effectiveness in drug synthesis.
FAQ's of Tamsulosin Hydrochloride IP BP:
Q: What is Tamsulosin Hydrochloride IP BP and what is its main application?
A: Tamsulosin Hydrochloride IP BP is a pharmaceutical intermediate in powder form, primarily used in the production of various medicine-grade pharmaceutical products. It serves as a building block in the synthesis of active pharmaceutical ingredients, particularly for urology medications.Q: How should Tamsulosin Hydrochloride IP BP be stored to ensure stability?
A: The powder should be stored at room temperature, as this condition preserves its chemical integrity and maximizes shelf life. Proper storage away from humidity and direct sunlight is essential for maintaining product quality.Q: What process is involved in using Tamsulosin Hydrochloride IP BP in pharmaceutical manufacturing?
A: Tamsulosin Hydrochloride IP BP acts as an intermediate in pharmaceutical synthesis. It undergoes further chemical reactions and processing steps before being incorporated into final medicinal formulations, especially those targeting urinary tract symptoms.Q: When is it advisable to utilize Tamsulosin Hydrochloride IP BP in pharmaceutical production?
A: It is suitable to be used at the stage where medicine-grade, high-purity intermediates are required during the synthesis of pharmaceuticals. Its quality makes it especially relevant for producing therapeutics that demand strict regulatory compliance.Q: Where can Tamsulosin Hydrochloride IP BP be sourced reliably in India?
A: You can obtain Tamsulosin Hydrochloride IP BP from reputable exporters, service providers, suppliers, and traders in India who specialize in pharmaceutical-grade intermediates and adhere to IP/BP purity standards.Q: What are the benefits of using medicine-grade Tamsulosin Hydrochloride IP BP?
A: Using our medicine-grade intermediate assures optimal purity, compliance with international pharmacopoeial standards, and reliability in pharmaceutical manufacturing processes, leading to higher-quality end products.Q: How is Tamsulosin Hydrochloride IP BP typically utilized by pharmaceutical manufacturers?
A: Manufacturers incorporate the powder intermediate into their chemical synthesis workflows, using it in the production of active pharmaceutical ingredients required for medications treating specific urological conditions.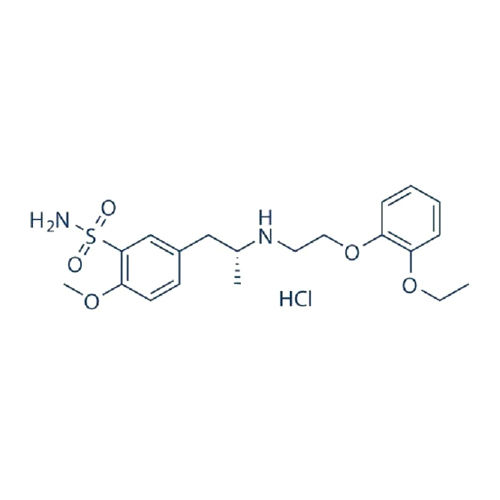
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Active Pharmaceutical Ingredients Category
Cefixime API Powder
Minimum Order Quantity : 50 Kilograms
Storage : Dry Place
Grade : Medicine Grade
Type : Pharmaceutical Intermediates
Physical Form : Powder
Mupirocin IP BP USP
Minimum Order Quantity : 10 Kilograms
Storage : Room Temperature
Grade : Medicine Grade
Type : Pharmaceutical Intermediates
Physical Form : Powder
Folic Acid Powder
Price 3900 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Storage : Room Temperature
Grade : Medicine Grade
Type : Pharmaceutical Intermediates
Physical Form : Powder
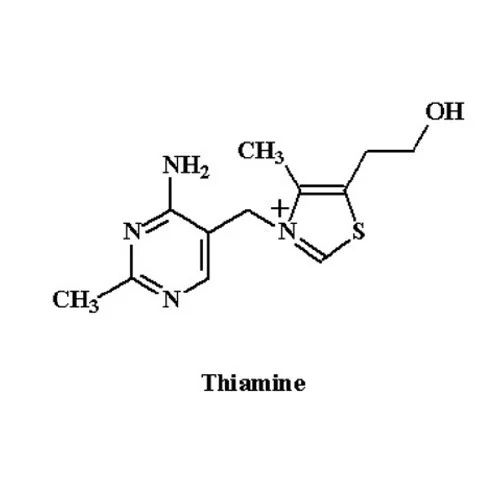
Thiamine Vitamin B1
Price 1975 INR / Kilograms
Minimum Order Quantity : 50 Kilograms
Storage : Room Temperature
Grade : Medicine Grade
Physical Form : Powder
We deal only in bulk quantity orders.
 Send Inquiry
Send Inquiry





 Send Inquiry
Send Inquiry Send SMS
Send SMS English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese